DC's Batman #35 by Snyder and Capullo
Batman gets in his big boy armor to be able to play with everyone else. Don't worry, Superman shows him a thing or two. However, his way of dealing with Aquaman is to spray him with an absorbent compound, magnesium sulfate, which will dry Mr. Landfish out.
Questions:
1.) Is magnesium carbonate an absorbing agent?
2.) Is it the worlds best absorbing agent?
3.) How do absorbing agents work?
Answers:
1.) Yes, magnesium carbonate (MgCO3) is used as a drying agent. It can hold 5 moles of water per 1 mole of magnesium carbonate.
Batman gets in his big boy armor to be able to play with everyone else. Don't worry, Superman shows him a thing or two. However, his way of dealing with Aquaman is to spray him with an absorbent compound, magnesium sulfate, which will dry Mr. Landfish out.
Questions:
1.) Is magnesium carbonate an absorbing agent?
2.) Is it the worlds best absorbing agent?
3.) How do absorbing agents work?
Answers:
1.) Yes, magnesium carbonate (MgCO3) is used as a drying agent. It can hold 5 moles of water per 1 mole of magnesium carbonate.
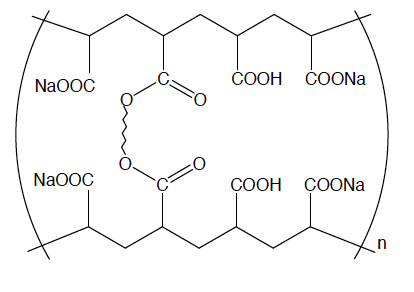
| 2.) No, magnesium carbonate is only a moderate strength drying agent. magnesium sulfate (MgSO4) is also an absorbing agent. It's strength is moderate by holding onto 7 moles of water compounds per 1 mole of magnesium sulfate. It is an acidic drying agent that works in many solvents However, sodium sulfate (Na2SO4) is better as it can hold 10 moles of water. A polymer, sodium polyacrylate (the compound on the right), is super absorbent. It can hold up to 800 times its weight in water which is the equivalent of about 1500 moles per one mole of sodium polyacrylate. |
3.) Magnesium carbonate and the sulfate compounds listed above work by allowing the water molecules to become part of the structure of the compound, which is called a hydrate. So, each molecule would have a certain number of water molecules attached to it. However, sodium polyacrylate works by osmosis. It absorbs the water and holds it in, however if the salt content is changed it cannot hold as much water. Considering that, it can hold 800 times its weight of distilled water but only about 300 times its weight of tap water since there are a lot of ions in it.
Sources:
http://www.chem.ucla.edu/~bacher/Specialtopics/Drying%20Agents.html
http://en.wikipedia.org/wiki/Magnesium_carbonate
http://ncsu.edu/project/chemistrydemos/Organic/Superabsorbent.pdf
Sources:
http://www.chem.ucla.edu/~bacher/Specialtopics/Drying%20Agents.html
http://en.wikipedia.org/wiki/Magnesium_carbonate
http://ncsu.edu/project/chemistrydemos/Organic/Superabsorbent.pdf


 RSS Feed
RSS Feed